pH is a term used to describe the relative amount of acidity or basicity in the wastewater. Low pH values indicate a high concentration of hydrogen ions (acids) in solution, and high pH values indicate a low concentration of hydrogen ions (basic). The pH value in water can range from 1 to 14.
There are two common times when a septic professional may be evaluating pH:
1. To evaluate treatment in a septic or pump tank or an advanced system
When evaluating a tank or treatment system a neutral pH of 6.5 to 7.5 is optimum for the microbes. The microbes that can survive outside the neutral range are not as efficient as the microbes that can survive at the neutral pH level. When opening a treatment system an odor may be noticeable indicating acidic or basic conditions. Low pH has a very acidic smell that absorbs readily into clothing and is hard to remove. High pH often smells like the chemical or cleaner that was used at the wastewater source and is causing the high pH. Over a relatively short period of time, our olfactory sensors become accustomed to an odor. As a result, the odor test can only be used at the very start of testing or inspection, before our senses get used to the odor.
Too high?
High pH (basic conditions) can be caused by certain laundry detergents, cleaning agents, chemicals, and high alkalinity source water. Photo developing labs, detergents, and soap-based products are often alkaline. Municipal water suppliers often raise the pH of drinking water to prevent corrosion of pipes, often to pH 9.
Too low?
Low pH (acidic conditions) can be influenced by low alkalinity in the water supply or acid-based cleaners. If there is an above-normal use of dairy products, soda, coffee, excessive baking, or home canning, lower pH levels in the wastewater stream are likely.  Nitrification can also cause a significant drop in pH if there is not adequate buffering capacity (alkalinity) in the wastewater. Because nitrification consumes about 7.1 mg of alkalinity (as CaCO3 ) for every mg of NH4+-N oxidized, in low alkalinity wastewaters there is a risk that nitrification will lower the pH to inhibitory levels.
Nitrification can also cause a significant drop in pH if there is not adequate buffering capacity (alkalinity) in the wastewater. Because nitrification consumes about 7.1 mg of alkalinity (as CaCO3 ) for every mg of NH4+-N oxidized, in low alkalinity wastewaters there is a risk that nitrification will lower the pH to inhibitory levels.
2. When liming septage to reduce pathogens and odors prior to land application
The purpose of adding lime to septage is to treat and reduce the number of pathogens present in the septage and to reduce odors. The high pH kills bacteria, viruses and parasites. Odor reduction occurs because the high pH slows the biological activity and break down of the septage. The requirement is to achieve a pH of 12 and hold it at 12 for 30 minutes. Another important measurement when taking the pH is the temperature of the septage. Because the solubility of lime changes with temperature, the pH that you measure will not be accurate unless you make a temperature correction. There is approximately a 0.03 change in pH for each degree change in temperature above or below 25° Celsius (77° Fahrenheit). Actual pH = Measured pH + [ 0.0167 x (Temp °F - 77) ]
MEASURING pH
The pH of the wastewater or septage can be measured using a pH meter and electrode, pH pocket meter or litmus paper.
Laboratory-quality pH meter and electrode
The pH meter and electrode, generally restricted to laboratory analysis of field-collected samples, offers the highest degree of accuracy and precision. The pH meter-electrode measures the electric potential, which is a function of the H+ activity in water samples. Calibration is completed with two buffer solutions.
Pocket meters
Pocket meters can be easily used in the field. Pocket meters and multi-parameter probes are electronic hand-held testers that are dipped directly into the water body and provide a digital readout of pH. Pocket pals are typically calibrated with one buffer. It must be calibrated in accordance with the manufacturer’s specification.
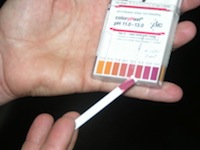
Color comparators/pH strips
With color comparators, you add a reagent to the water sample that colors the sample for a visual comparison. pH strips are dipped into water samples and then change color according to the pH. The intensity of the color is proportional to the pH in the sample, and colors are compared on a chart. You should use litmus paper with a pH range that is expected. For wastewater a range of 4 to 10 will be appropriate; septage should be between 10 and 14. The paper should have a sensitivity that can measure changes in pH at 0.1 increments.
About the Author
Sara Heger, Ph.D., is an engineer, researcher and instructor in the Onsite Sewage Treatment Program in the Water Resources Center at the University of Minnesota. She presents at many local and national training events regarding the design, installation and management of septic systems and related research. Heger is education chair of the Minnesota Onsite Wastewater Association (MOWA) and the National Onsite Wastewater Recycling Association (NOWRA), and serves on the NSF International Committee on Wastewater Treatment Systems. Send her questions about septic system maintenance and operation by email to kim.peterson@colepublishing.com.