Although the atmosphere around us is 78% nitrogen, various forms of nitrogen can cause problems in the incorrect location and form, causing both public health and environmental concerns.
The most commonly cited health concern is when groundwater used as a drinking water source contains elevated levels of nitrogen which can cause methemoglobinemia, a condition that occurs in infants, especially those less than six months old. After ingestion, nitrate is reduced to nitrite in the gut of the infant. The absorbed nitrite reacts with hemoglobin in the blood, forming methemoglobin. Methemoglobin, unlike hemoglobin, cannot carry oxygen. As more of the blood hemoglobin is converted to methemoglobin, the oxygen-carrying capacity of the blood is significantly reduced.
When excess nitrogen concentrations are discharged to surface waters, it can also cause environmental problems. Nitrogen is sometimes the limiting nutrient for the growth of algae and aquatic plants in surface waters, particularly coastal or ocean waters. Excess nitrogen can negatively impact these waters with odors, accumulation of biomass, dissolved oxygen depletion due to biomass decay and loss of fish and shellfish. The oxidation through the process of nitrification can exert a significant oxygen demand on the receiving water with the nitrogen-related oxygen demand sometimes higher than the carbonaceous biochemical oxygen demand. Lastly, nitrogen in the form of ammonia can cause acute toxicity to species of fish.
When ground or surface water have elevated levels of nitrogen, we must look at all the sources to determine which sources may be contributing significantly to the problem and then direct efforts and resources where they make the most sense.
Agricultural Sources
Agricultural activities are a significant source of nitrate in ground and surface waters. Nitrate can enter groundwater at elevated levels by excessive or inappropriate use of nitrogen-containing nutrient sources, which include commercial fertilizers and animal manure. Even with improved nutrient management the use of nitrogen fertilizer continues to increase, and it is the largest source of nitrate contamination of groundwater in the U.S. Most nitrogen fertilizer is applied as anhydrous ammonia, urea, or as nitrate or ammonium salt. In an aerobic soil environment, applied ammonia can be transformed to nitrate which readily migrates to groundwater through most soil types.
Livestock and dairy practices that concentrate animals can also significantly contribute to nitrate contamination of groundwater if the animal wastes generated by the operation are not effectively managed. The types of crops and cropping system are also important in determining the potential for nitrate migration to groundwater. Irrigated agriculture on sandy soils, and heavily fertilized, shallow-rooted crops, favor nitrate leaching.
Wastewater Sources
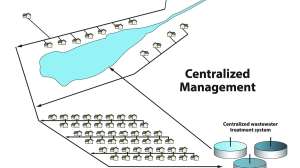
Contamination of ground and surface waters with nitrates from septic systems is a problem in parts of the U.S. When there is discussion about outdated septic systems, nitrate in groundwater or impacts to coastal waters is often a topic of concern. The annual nitrogen contribution for a family of four from a conventional septic system on a quarter-acre lot is approximately 50 pounds per year while the annual nitrogen requirement for a quarter acre of Bermuda grass is also about 50 pounds per year. The problem, however, is that the nitrogen from septic systems is not uniformly distributed throughout a lawn and is sometimes discharged at a depth below which plants can utilize it.
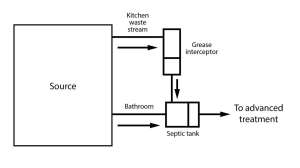
Nitrogen primarily exists in septic tank effluent as Total Kjeldahl nitrogen which is the combination of organic nitrogen and ammonia/ammonium. As the TKN moves through the aerobic environment in the soil it is usually transformed into nitrate. Due to its negative charge nitrate tends not to be held by the soil but moves the effluent. There are modifications that can be made to conventional systems to reduce this risk including shallow or surface application and advanced systems which can remove the nitrogen through denitrification.
Atmospheric Deposition Sources
Atmospheric deposition may be in a wet form as rain, snow, hail, fog, and freezing rain or in a dry form as particulates, gases and droplets. The largest sources are point sources — coal- and oil-burning electric utilities and large industries that together account for over 50% of nitrogen emissions. However, automobiles, trucks, buses and other forms of transportation account for approximately 38% of nitrogen emissions.
Stormwater Sources
Fertilizers, yard and pet waste and certain soaps and detergents contain nitrogen and can contribute to nutrient pollution if not properly used or disposed of. The amount of hard surfaces and type of landscaping can also increase the runoff of nitrogen during wet weather. When precipitation falls on our cities and towns it runs across hard surfaces such as rooftops, sidewalks and roads and carries nitrogen into local waterways.
Fixation Sources
Biological fixation is the most common method of fixation (legumes like soybeans do this), but it can also occur by lightning where nitrogen in the air is converted into nitrate.
About 40 million lightning strikes hit the ground in the United States each year. Lightning can reach temperatures of 50,000 degrees and up to a billion volts of electricity. When it strikes, it separates the bonds in the airborne nitrogen molecules which quickly bond to oxygen in the atmosphere. Along with the lightning in the cloud are droplets and raindrops. Nitrogen dioxide dissolves in water, creating nitric acid, which forms nitrates. The nitrates fall to the ground in raindrops and seep into the soil in a form that can be absorbed by plants but due to the negative charge can flow into ground and surface waters. There can be 6-12 pounds of nitrate created per strike. In some areas, this naturally occurring form of nitrate may not be significant but if you live in an area with low inputs from agriculture and human waste it should be considered.
There is data to suggest our current weather patterns are making thunderstorms more intense, increasing the amount of lightning, and therefore the amount of nitrogen being produced. This also has the risk of igniting more wildfires.
Source Variations
The proportions of sources of nitrogen vary from watershed to watershed throughout the US. These proportions vary as a function of many factors such as land use, geology and soil, population, hydrologic conditions, and climate. We should try to reduce inputs, when possible, but this variability suggests we need watershed specific plans to address problems. A more complete understanding of which sources have the greatest effects is essential to implementing effective programs.